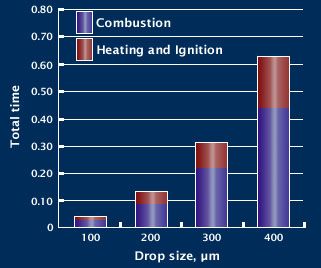
Emissions
Problem
 Incomplete combustion produces carbon monoxide, soot particles and other emissions that are harmful to the environment.
Incomplete combustion produces carbon monoxide, soot particles and other emissions that are harmful to the environment.
Fuel oil contains varying amounts of sulfur, which forms sulfur dioxide during combustion and sulfur trioxide when there is sufficient oxygen available in the plant or as the emissions leave the stack.
Sulfur trioxide, when allowed to cool below its dew-point, will form sulfuric acid, an extremely corrosive substance that causes severe damage to both boiler plants and diesel engines.
The emission of SO₃ causes acid rain, an environmental nightmare, along with other environmental problems.
NOx is formed when O₂ levels are raised to improve combustion. NOx also creates serious environmental problems.
Solution
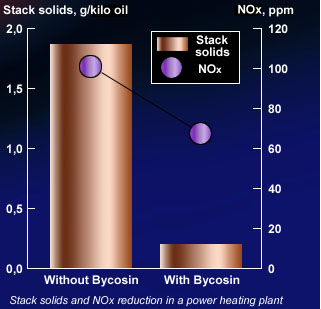 Reduce the formation of SO₃ and NOx.
Reduce the formation of SO₃ and NOx.
Reduce the environmental impact by reduction of the SO₃ and NOx emissions.
Prevent formation of vanadium deposits, which reduces the catalytic oxidation of SO₂ to SO₃.
Provide better plant economy through lower environmental charges.
Particulates
Problem
 Particulates are made up of a number of things, including unburned carbon, metallic ash and other materials that are in the fuel but are not consumed during combustion.
Particulates are made up of a number of things, including unburned carbon, metallic ash and other materials that are in the fuel but are not consumed during combustion.
Particulates can also come from the addition of any material that would add to the ash loading of the fuel, such as inorganic magnesium that a plant may add to the fuel to reduce SO₃ corrosion.
Particulates foul the environment, form deposits and cause fouling throughout the plant. Oil consumption goes up and power output down. Other effects are more wear and damage to the system and more downtime for cleaning.
Solution
Incendio products:
Catalyse the combustion thus reducing the amount of soot and coke in the flue gases.
Improve the atomization of the fuel. The smaller droplets produced provide a better mixing with oxygen giving earlier completion of combustion.
Shorten the flame by making the fuel burn faster, which reduces soot formation.
Disperse asphaltenes which allows a better spray pattern and more complete combustion.
Reduce the soot number, which allows a reduction in excess air, resulting in both lower NOx and less SO₃ in the combustion gases.
Reduce soot and stack solids emissions.
Prevent soot and coke deposits.
Opacity
Problem
 Opacity is a visual, environmental and health issue and is causing both local and national governments to enact strict laws governing allowed levels.
Opacity is a visual, environmental and health issue and is causing both local and national governments to enact strict laws governing allowed levels.
Opacity is usually measured in the stack, which may give a very different number than what is seen in the plume coming from the top of the stack.
There are three things that impact visible opacity; incomplete combustion, SO₃ and NOx. While a plant may be keeping its stack opacity within limits, the visible plume may result in complaints from the neighborhood around the plant.
Lowering SO₃ can only be done by putting in scrubbers, a very expensive process, or reducing the sulfur in the fuel which is now required in many European countries.
SO₃ can be more problematic and visible during a “rampup”_at a power station, when sulfuric acid, which condensed over night, is evaporated and leaves the stack in heavy concentrations.
NOx is both hazardous to the environment and a controlled emission that impacts opacity. Opacity may also only be steam but most people are not aware of that.
Solution
 Both opacity and the visible plume can be reduced by use of Incendio additives.
Both opacity and the visible plume can be reduced by use of Incendio additives.
The combination of Incendio catalyst and organic magnesium makes it possible to have a better and more stable combustion.
The catalyst allows a reduction in excess air, reduces unburned carbon, CO and formation of NOx and SO₃. Formed SO₃ will be neutralised by the organic magnesium. Less SO₃ in the flue gases will give a less visible plume from the stack.
Incendio additives clean sodium and vanadium deposits from boiler walls and tubes. Vanadium is known as a catalyst of SO₃ formation so reduced deposits means less corrosion.
Corrosion
Problem
 Corrosion is a severe problem that can impact all parts of the combustion system from injection to stack emissions.
Corrosion is a severe problem that can impact all parts of the combustion system from injection to stack emissions.
Corrosion is caused by the sulfur in the fuel and generally more sulfur in the fuel creates corrosion problems.
Sulfur is burned during combustion forming SO₂ (sulfur dioxide) that is not a corrosive gas. It’s only when oxygen is available to the SO₂ that it becomes SO₃ and can cause both hot and cold end corrosion.
Vanadium is another contributor to corrosion. If vanadium reaches its melting temperature in the boiler or engine it creates a deposit on the metal surface. Between the deposit and the metal, sulfuric acid is formed and corrodes the metal.
Corrosion can cause huge expenses to a company in spare parts, down time and hourly labor.
Solution
 The best solution to this problem is to buy fuel without sulfur. Trusting that this is not a reasonable solution for most companies the next best option is to add some form of magnesium to the combustion so that the melting temperature of the vanadium is raised high enough to leave the vanadium as an ash and not a deposit.
The best solution to this problem is to buy fuel without sulfur. Trusting that this is not a reasonable solution for most companies the next best option is to add some form of magnesium to the combustion so that the melting temperature of the vanadium is raised high enough to leave the vanadium as an ash and not a deposit.
The magnesium breakes the vanadium bond in existing deposits allowing them to break off the metal surface, improving heat transfer and reducing corrosion.
The magnesium will reduce corrosion in the back end of the furnace that could be prone to SO₃ condensation. A key issue here is what magnesium to use. Incendio recommends organometallic magnesium.
Deposits
Problem
 Deposits come from poor combustion, unburnt carbon and the ash in the fuel.
Deposits come from poor combustion, unburnt carbon and the ash in the fuel.
Deposits reduce efficiency, increase particulate emissions, increase maintenance costs and can lead to expensive and unnecessary down time.
The combustion of fuel oil with high vanadium, sulfur and sodium content often result in corrosive deposits.
Deposits are built up when reaction products from these compounds melt and are deposited on hot surfaces. These deposits can be very hard to remove.
Vanadium’s melting temperature changes with the presence of sodium in the fuel.
The typical rule of thumb is that when the ratio between sodium and vanadium is 1:3, the melting temperature of the created deposits will drop significantly and result in sticky deposits that easily adhere to all surfaces.
Today, due to the increase of asphaltene content in the fuel, high MCR and generally poor quality fuel, the likely hood of deposits is greatly increased.
Solution
 In order to prevent or reduce deposits and corrosion, the melting point for the sodium and vanadium salts must be raised.
In order to prevent or reduce deposits and corrosion, the melting point for the sodium and vanadium salts must be raised.
A higher melting point means drier and more brittle deposits that do not adhere to the metal surfaces, are easy to remove or will be carried trough the boiler and out through the stack as a light ash.
Using oil soluble organometallic magnesium, at the right ppm, can raise vanadium’s melting temperature, keeping it from forming a deposit.
The magnesium will also break the vanadium bond in the existing deposits allowing them to break off the metal surface, improving heat transfer and reducing corrosion.
The magnesium will reduce corrosion in the back end of the furnace that could be prone to SO₃ condensation.
With carbon deposits, the first step is to be sure that the plant is operating correctly. If that is not the problem, then poor combustion of the fuel is and it can be improved using an oil soluble organometallic in the correct ppm. Incendio has a number of products that improve combustion and injection systems that mix those products with the fuel in the correct proportions.
Incendio products eliminate deposits. Incendio also continously improves its products to make them more effective and less expensive to the user.
The Incendio products:
• Raise the melting point of vanadium salts, thus preventing deposits.
• Make existing deposits brittle and easy to remove.
• Allow better heat transfer by maintaining the cleanliness of the superheater tubes of the boiler.
• Prevent corrosion of the tubes.
• Eliminate carbon deposits through improved combustion.
• Are produced with very high concentrations of organometallics, making them very effective at low dosages.
• Reduce maintenance costs.
Fuel Stability
Problems
 Fuel stability is becoming an increasing problem for those working with heavy fuels. When fuels from different crude sources are blended, the fuel often becomes unstable and difficult to burn.
Fuel stability is becoming an increasing problem for those working with heavy fuels. When fuels from different crude sources are blended, the fuel often becomes unstable and difficult to burn.
The fuel may also stratify and be difficult to heat and inject at the correct temperatures.
Fuel today is also much higher in asphaltenes that tend to agglomerate and precipitate out to form sludge which can foul the fuel system, plugging fuel lines, heaters, purifiers, viscometers, injectors or burner tips.
The asphaltenes can also negatively impact combustion by making the fuel harder to burn. Fuel that has more than 6% asphaltenes will result in retarded ignition and poor combustion characteristics.
Solution
 Today, it is becoming common practice to use a fuel dispersant and stabilizer to reduce problems related to fuel blending and high asphaltene content.
Today, it is becoming common practice to use a fuel dispersant and stabilizer to reduce problems related to fuel blending and high asphaltene content.
Incendio has a number of products on the market that will improve fuel stability and reduce asphaltene agglomeration as well as remove sludge from the fuel system.
Benefits

Incendio products will:
• Reduce soot and stack solids emissions.
• Reduce corrosion and corrosive deposits.
• Result in fewer operational stoppages for furnace cleaning.
• Give higher effiency through better heat transfer.
• Allow more complete combustion for better fuel economy.
• Allow the possibility to mix fuels of different qualities.
• Disperse sludge which is burned with the oil.
• Disperse asphaltenes throughout the fuel.
• Reduce piston rings and cylinder liner wear.
• Extend the time between engine overhauls.
• Reduce soot emissions.